

发电技术 ›› 2026, Vol. 47 ›› Issue (2): 248-256.DOI: 10.12096/j.2096-4528.pgt.260202
• 发电及环境保护 • 上一篇
姚晨翔1, 王争荣2, 夏芝香1, 李超1, 孙路长2, 王凯亮2, 胡昔鸣3, 方梦祥1
收稿日期:2025-02-24
修回日期:2025-05-10
出版日期:2026-04-30
发布日期:2026-04-21
作者简介:基金资助:Chenxiang YAO1, Zhengrong WANG2, Zhixiang XIA1, Chao LI1, Luchang SUN2, Kailiang WANG2, Ximing HU3, Mengxiang FANG1
Received:2025-02-24
Revised:2025-05-10
Published:2026-04-30
Online:2026-04-21
Supported by:摘要:
目的 化学吸收剂稳定性是化学吸收碳捕集系统稳定运行的关键,为此,研究新型混合胺吸收剂BA-1氧化降解和热降解问题,并进行抗降解剂研究。 方法 通过热降解实验装置,在不同温度(100、120、140 ℃)和CO2负载浓度(0~1.5 mol/L)条件下,对BA-1吸收剂的热降解行为进行了研究。同时,利用氧化降解实验装置,在65 ℃、纯氧氛围下考察了BA-1吸收剂的氧化降解性能。通过阳离子色谱和阴离子色谱技术,分析了BA-1吸收剂的降解率及降解产物。并对2,5-二巯基-1,3,4-噻二唑(DMTD)、螯合剂E-1、Na2SO3和乙醛肟(AAO)的抗氧化作用进行了测试。 结果 BA-1吸收剂的降解主要由氧化降解引起。在添加E-1、Na2SO3和AAO后,BA-1吸收剂的降解速率得到有效抑制。其中,添加质量分数0.3%的E-1时,BA-1在24天内的降解率仅为22.5%,降解抑制率高达53.8%。降解产物主要包括甲酸根、醋酸根、草酸根和硝酸根等物质,其中氧化降解产物的生成量是热降解的10倍以上,进一步证实了氧化降解是BA-1吸收剂降解的主要机制。 结论 混合胺吸收剂的降解主要是由氧化降解导致,添加螯合剂能较好地解决吸收剂在工业应用时的降解问题。
中图分类号:
姚晨翔, 王争荣, 夏芝香, 李超, 孙路长, 王凯亮, 胡昔鸣, 方梦祥. 燃煤烟气碳捕集混合胺吸收剂降解性能研究[J]. 发电技术, 2026, 47(2): 248-256.
Chenxiang YAO, Zhengrong WANG, Zhixiang XIA, Chao LI, Luchang SUN, Kailiang WANG, Ximing HU, Mengxiang FANG. Study on Degradation Performance of Mixed Amine Absorbent for Carbon Capture From Coal-Fired Flue Gas[J]. Power Generation Technology, 2026, 47(2): 248-256.
| 化学试剂 | 分子结构 |
|---|---|
| 乙醇胺(MEA) |  |
| 羟乙基乙二胺(AEEA) | 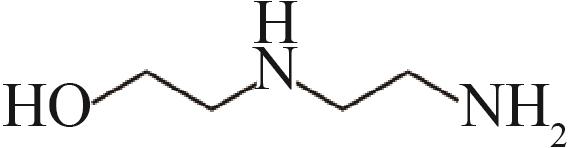 |
2,5-二巯基-1,3,4-噻二唑 (DMDT) | 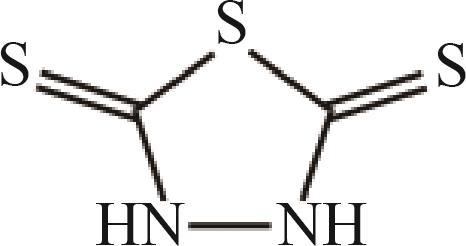 |
| 亚硫酸钠(Na2SO3) | 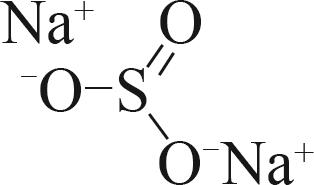 |
| 乙醛肟(AAO) |  |
| 四水合氯化亚铁(FeCl2·4H2O) | 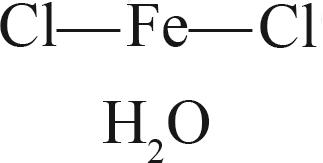 |
表1 实验用化学试剂
Tab. 1 Chemical reagents used in experiment
| 化学试剂 | 分子结构 |
|---|---|
| 乙醇胺(MEA) |  |
| 羟乙基乙二胺(AEEA) |  |
2,5-二巯基-1,3,4-噻二唑 (DMDT) |  |
| 亚硫酸钠(Na2SO3) |  |
| 乙醛肟(AAO) |  |
| 四水合氯化亚铁(FeCl2·4H2O) |  |
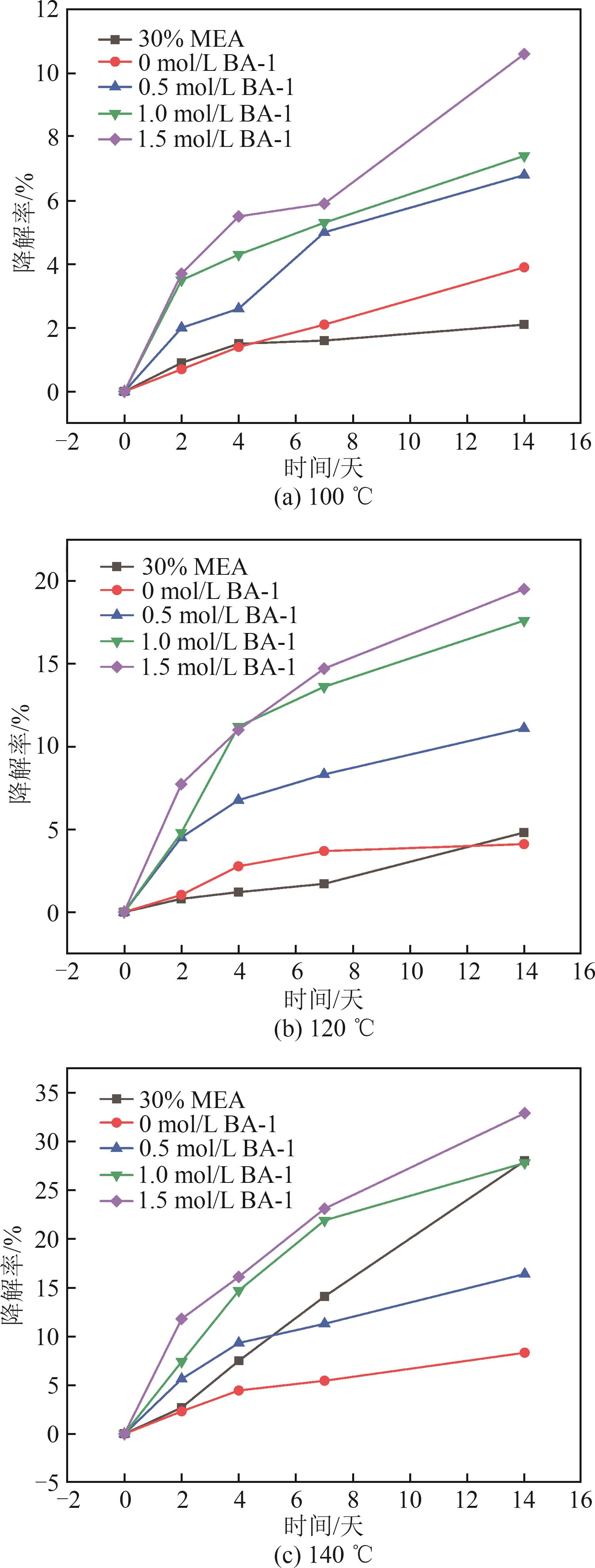
图3 在100、120、140 ℃下不同CO2负载的BA-1降解率随时间变化趋势
Fig. 3 Variation trends of BA-1 degradation rate with time under different CO2 loadings at 100 ℃, 120 ℃, and 140 ℃
| [1] | DUNN R, BLANNIN J, GOBRON N,et al .State of the climate in 2023[J].Bulletin of the American Meteorological Sociaty,2024,105(8):1-50. doi:10.1175/2025bamsstateoftheclimate.1 |
| [2] | 高凤玲,崔国民,陶乐仁,等 .全球变暖背景下温室效应的光谱能量分布分析[J].计算物理,2014,31(3):343-350. doi:10.3969/j.issn.1001-246X.2014.03.010 |
| GAO F L, CUI G M, TAO L R,et al .Spectral energy distribution analysis of greenhouse effect under the background of global warming[J].Computational Physics,2014,31(3):343-350. doi:10.3969/j.issn.1001-246X.2014.03.010 | |
| [3] | IEA .CO2 Emissions in 2023[EB/OL].(2024-03-01)[2025-01-12].. |
| [4] | IEA .CO total emissions by region,2000-2023[EB/OL].(2024-02-27)[2025-01-12].. |
| [5] | 于强 .CO2捕集与封存(CCS)技术现状与发展展望[J].能源与环境,2010(1):64-65. doi:JournalArticle/5af5ba1fc095d718d8288007 |
| YU Q .Current status and development prospects of CO2 capture and storage (CCS) technology[J].Energy and Environment,2010(1):64-65. doi:JournalArticle/5af5ba1fc095d718d8288007 | |
| [6] | 李彦斌,徐艺璇,葛睿,等 .考虑碳市场及合作博弈的GU-P2G-CCUS系统运行优化模型[J].电测与仪表,2025,62(7):1-10. |
| LI Y B, XU Y X, GE R,et al .An optimal GU-P2G-CCUS system operating model considering the carbon market and the cooperative game among participants[J].Electrical Measurement & Instrumentation,2025,62(7):1-10. | |
| [7] | 刘贵洲,方瑞瑞,窦立荣,等 .中国CCS/CCUS发展瓶颈与应对策略[J/OL].天然气与石油,1-14 [2024-10-20]. doi:10.3969/j.issn.1006-5539.2024.05.002 |
| LIU G Z, FANG R R, DOU L R,et al .Bottlenecks and countermeasures in the development of CCS/CUS in China[J/OL].Natural Gas and Petroleum,1-14 [2022-10-20]. doi:10.3969/j.issn.1006-5539.2024.05.002 | |
| [8] | 檀勤良,贺嘉明,吕函谕,等 .考虑成本不确定性的发电企业低碳技术采纳决策优化研究[J].中国电力,2025,58(5):62-73. doi:10.11930/j.issn.1004-9649.202403055 |
| TAN Q L, HE J M, LV H Y,et al .Optimization of low-carbon technology adoption decision for generation enterprises considering cost uncertainty[J].Electric Power,2025,58(5):62-73. doi:10.11930/j.issn.1004-9649.202403055 | |
| [9] | 张程,曾崟琳,匡宇 .考虑需求响应和碳捕集电厂灵活运行方式的综合能源系统调度[J].智慧电力,2024,52(9):88-95. |
| ZHANG C, ZENG Y L, KUANG Y .Integrated energy system scheduling considering demand response and flexible operation of carbon capture power plant[J].Smart Power,2024,52(9):88-95. | |
| [10] | 邬凯浪,单兰晴,薄利明,等 .考虑电力系统灵活爬坡需求的火电厂碳捕集改造方案优化方法[J].电力建设,2025,46(9):27-41. |
| WU K L, SHAN L Q, BO L M,et al .Optimization methodology for carbon capture reformation schemes in thermal power plants considering the power flexible ramping demand[J].Electric Power Construction,2025,46(9):27-41. | |
| [11] | 邢耀敏,高春雨,廖丛林 .考虑碳捕集及碳交易的虚拟电厂多目标优化调度[J].分布式能源,2025,10(3):42-52. doi:10.16513/j.2096-2185.DE.24090507 |
| XING Y M, GAO C Y, LIAO C L .Multi-objective optimization dispatching for virtual power plants considering carbon capture and carbon trading strategy[J].Distributed Energy,2025,10(3):42-52. doi:10.16513/j.2096-2185.DE.24090507 | |
| [12] | 王建行,赵颖颖,李佳慧,等 .二氧化碳的捕集、固定与利用的研究进展[J].无机盐工业,2020,52(4):12-17. |
| WANG J H, ZHAO Y Y, LI J H,et al . Research progress on capture, fixation and utilization of carbon dioxide[J].Inorganic Salt Industry,2020,52(4):12-17. | |
| [13] | MA’MUM S, SVENDSEN H F, HOFF K A,et al .Selection of new absorbents for carbon dioxide capture[J].Greenhouse Gas Control Technologies,2005,1:45-53. doi:10.1016/b978-008044704-9/50006-9 |
| [14] | STRAZISAR B R, ANDERSON R R, WHITE C M .Degradation pathways for monoethanolamine in a CO2 capture facility[J].Energy & Fuels,2003,17(4): 1034-1039. doi:10.1021/ef020272i |
| [15] | GOTO K, YOGO K, HIGASHII T .A review of efficiency penalty in a coal-fired power plant with post-combustion CO2 capture[J].Applied Energy,2013,111:710-720. doi:10.1016/j.apenergy.2013.05.020 |
| [16] | 孙宇航,李超,王争荣,等 .甲基二乙醇胺-二元胺混合体系烟气CO2吸收再生性能研究[J].发电技术,2024,45(3):468-477. |
| SUN Y H, LI C, WANG Z R,et al .Study on the CO2 absorption and regeneration performance of the mixed system of methyl diethanolamine and diamine in flue gas[J].Power Generation Technology,2024,45(3):468-477. | |
| [17] | LI G, SHEN X, JIAO X,et al .Novel tri-solvent amines absorption for flue gas CO2 capture:efficient absorption and regeneration with low energy consumption[J].Chemical Engineering Journal,2024,493:152699. doi:10.1016/j.cej.2024.152699 |
| [18] | WAI S K, SAIWAN C, IDEM R .Carbon dioxide (CO2) solubility in diethylenetriamine and 2-Amino-2-Methyl-1-Proponal (DETA-AMP) solvent system for amine-based CO2 capture in flue gas from coal combustion[C]//International Conference on Greenhouse Gas Control Technologies,2017. doi:10.1016/j.egypro.2017.03.1329 |
| [19] | 徐冬,黄艳,宋俊朝,等 .低能耗CO2混合胺吸收剂复配筛选与工业验证[J].洁净煤技术,2024,30(8):90-98. |
| XU D, HUANG Y, SONG J C,et al .Screening and industrial validation of low-energy CO2 mixed amine absorbent compound[J].Clean Coal Technology,2024,30(8):90-98. | |
| [20] | 陈阿小,冉真真,马双忱,等 .火电厂碳捕集技术:现状、应用与发展[J].洁净煤技术,2024,30(S1):52-64. |
| CHEN A X, RAN Z Z, MA S C,et al .Carbon capture technology in thermal power plants:current status, application, and development[J].Clean Coal Technology,2024,30(S1):52-64. | |
| [21] | 向国育,申长俊,陆诗建,等 .二氧化碳捕集、利用与封存示范工程进展[J].低碳化学与化工,2025,50(3):113-122. doi:10.12434/j.issn.2097-2547.20240202 |
| XIANG G Y, SHEN C J, LU S J,et al .Progress of carbon dioxide capture, utilization, and storage demonstration projects[J].Low Carbon Chemistry and Chemical Engineering,2025,50(3):113-122. doi:10.12434/j.issn.2097-2547.20240202 | |
| [22] | 杨清泉,杨德锋,郝金军,等 .一乙醇胺在烟气 CO2 回收系统中的降解及对策[J].化学工业与工程技术, 2005,26(2):53-54. doi:10.3969/j.issn.1006-7906.2005.02.018 |
| YANG Q Q, YANG D F, HAO J J,et al .Degradation and countermeasure of mono-ethanolamine in reclaim system of CO2 in flue gas[J].Journal of Chemical Industry & Engineering,2005,26(2):53-54. doi:10.3969/j.issn.1006-7906.2005.02.018 | |
| [23] | REYNOLDS A J, VERHEYEN T V, MEULEMAN E .Degradation of amine-based solvents[M].Amsterdam:Elsevier,2016:399-423. doi:10.1016/b978-0-08-100514-9.00016-0 |
| [24] | YOON B, STOWE H M, HWANG G S .Molecular mechanisms for thermal degradation of CO2-loaded aqueous monoethanolamine solution:a first-principles study[J].Physical Chemistry Chemical Physics,2019,21(39):22132-22139. doi:10.1039/C9CP04518J |
| [25] | BEDELL S A .Oxidative degradation mechanisms for amines in flue gas capture[J].Energy Procedia,2009,1(1):771-778. doi:10.1016/j.egypro.2009.01.102 |
| [26] | DU J, YANG W, XU L,et al .Review on post-combustion CO2 capture by amine blended solvents and aqueous ammonia[J].Chemical Engineering Journal,2024,488:150954. doi:10.1016/j.cej.2024.150954 |
| [27] | LIU H, NAMJOSHI O A, ROCHELLE G T .Oxidative degradation of amine solvents for CO2 capture[J].Energy Procedia,2014,63:1546-1557. doi:10.1016/j.egypro.2014.11.164 |
| [28] | JØRUND E .Screening of inhibitors for amine degradation[D].Trondheim:Norwegian University of Science and Technology,2012. |
| [29] | 黄宇辉,方梦祥,王涛,等 .新型MEA-AMP混合胺吸收剂的抗降解剂研究[J].能源工程,2021(5):35-42. |
| HUANG Y H, FANG M X, WANG T,et al .Inhibitor for new MEA-AMP blended amine absorbent[J]. Energy Engineering,2021(5):35-42. | |
| [30] | 储可弘,陈绍云,李强,等 .基于N-乙基乙醇胺非水CO2吸收剂的抗氧化剂[J].化工进展,2019,38(12):5565-5571. doi:10.16085/j.issn.1000-6613.2019-0403 |
| CHU K H, CHEN S Y, LI Q,et al .Antioxidant based on N-Ethanolamine non aqueous CO2 absorbent [J].Chemical Progress,2019,38 (12):5565-5571. doi:10.16085/j.issn.1000-6613.2019-0403 | |
| [31] | WANG Y W, FANG M X, GAO J,et al .Corrosion performance of A3 carbon steel in 30wt.% MEA and AMP-MEA blends for post-combustion carbon capture: effect and mechanism of corrosion inhibitors[J].International Journal of Greenhouse Gas Control,2024,136:104186. doi:10.1016/j.ijggc.2024.104186 |
| [1] | 朱法华, 徐静馨, 沈凡卉, 张西, 李辉, 田文鑫, 赵秀勇, 柏源. 中国火电低碳发展现状及展望[J]. 发电技术, 2026, 47(2): 237-247. |
| [2] | 邵雁, 谢颖, 刘子豪, 方梦祥, 许晓明, 胡昔鸣, 夏阳, 陈伟, 朴永玉. 钢铁行业碳捕集与利用技术研究进展[J]. 发电技术, 2026, 47(1): 1-13. |
| [3] | 王秀云, 章婉钰. 考虑电转气-碳捕集电厂联动的综合能源系统㶲经济分析[J]. 发电技术, 2026, 47(1): 14-25. |
| [4] | 罗城鑫, 吕彦龙, 刘润宝, 谢玉荣, 王雨昊, 刘锋, 隋军. 基于热泵技术的低碳排冷热电联供系统㶲经济性能研究[J]. 发电技术, 2025, 46(6): 1085-1096. |
| [5] | 路诗梦, 孙建林, 曾凡杰, 林小杰, 吴均湛, 马添翼, 钟崴, 谢立坤, 谢伟. 零碳地热能综合利用技术研究进展[J]. 发电技术, 2025, 46(5): 909-922. |
| [6] | 杨龙, 郝黎明, 寻志伟, 顾永正, 于芳, 邓博文, 高腾飞. Mo掺杂石墨烯催化CO2加氢制甲醇的机理研究[J]. 发电技术, 2025, 46(5): 923-929. |
| [7] | 马恺, 袁至, 李骥. 考虑氢能多元化利用的综合能源系统低碳经济调度[J]. 发电技术, 2025, 46(2): 263-273. |
| [8] | 胡昔鸣, 董文峰, 王争荣, 孙路长, 王凯亮, 李超, 方梦祥, 李治甫. 二氧化碳捕集塔内熔融共混亲水改性聚丙烯规整填料性能研究[J]. 发电技术, 2025, 46(2): 296-303. |
| [9] | 黄忠源, 金绪良, 孟凡钦, 殷爱鸣, 张丽, 陈绍云. 天然气电厂新型多元胺基CO2吸收剂试验与应用研究[J]. 发电技术, 2025, 46(2): 304-313. |
| [10] | 胡山鹰, 金涌, 张臻烨. 发展新质生产力,实现碳中和[J]. 发电技术, 2025, 46(1): 1-8. |
| [11] | 单思珂, 刘含笑, 刘美玲, 王帅, 崔盈. 我国火电行业碳足迹评估综述[J]. 发电技术, 2024, 45(4): 575-589. |
| [12] | 赵振宇, 包格日乐图, 李炘薪. 基于信息间隙决策理论的含碳捕集-电转气综合能源系统优化调度[J]. 发电技术, 2024, 45(4): 651-665. |
| [13] | 袁鑫, 刘骏, 陈衡, 潘佩媛, 徐钢, 王修彦. 碳捕集技术应用对燃煤机组调峰能力的影响[J]. 发电技术, 2024, 45(3): 373-381. |
| [14] | 孙宇航, 李超, 王争荣, 孙路长, 王凯亮, 胡昔鸣, 方梦祥, 张锋. 甲基二乙醇胺-二元胺混合体系烟气CO2吸收再生性能研究[J]. 发电技术, 2024, 45(3): 468-477. |
| [15] | 龚思琦, 云再鹏, 许明, 敖乐, 李初福, 黄凯, 孙晨. 基于三元催化剂的固体氧化物燃料电池尾气催化燃烧数值模拟[J]. 发电技术, 2024, 45(2): 331-340. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||